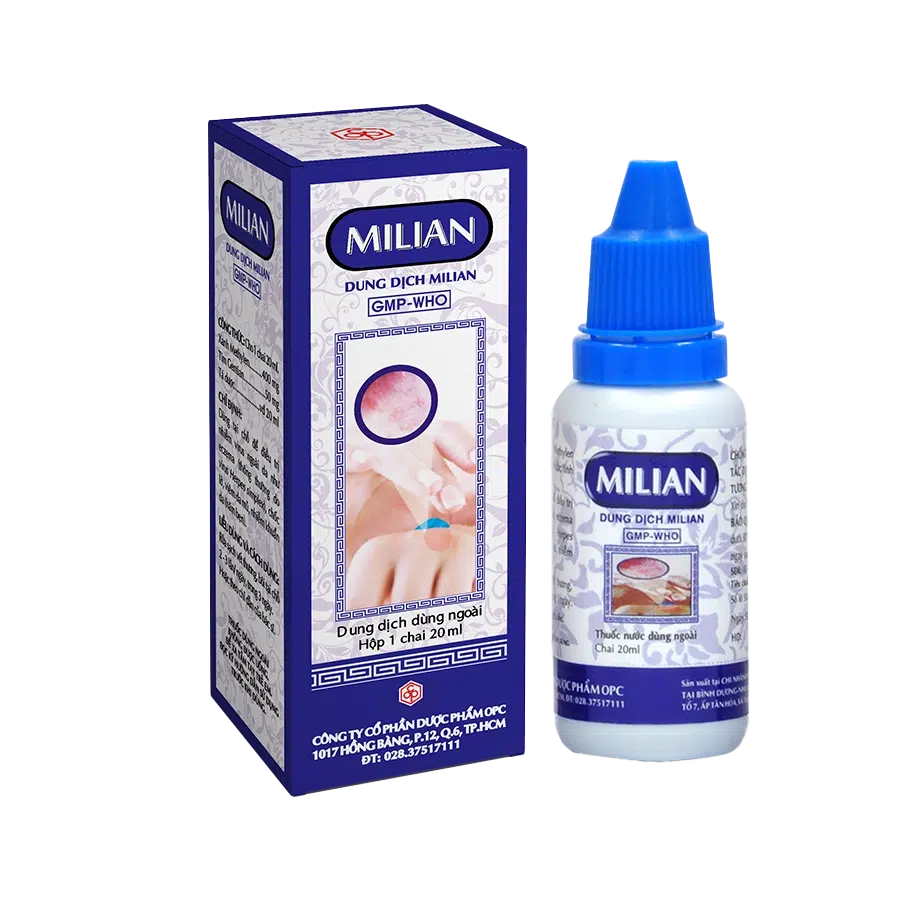
MILIAN SOLUTION
FORMULA:
For 1 bottle of 20 ml.
Active ingredient:
Methylene blue 400 mg; Gentian violet 50 mg
Excipients: Purified water
INDICATIONS:
Topical use for the treatment of skin viral infections such as eczema (usually caused by Herpes simplex virus), impetigo, pyoderma, skin infections (inguinal rash).
DOSAGE AND ADMINISTRATION:
Wash the wound, apply topically 2-3 times / day, for 3 days.
CONTRAINDICATIONS:
Open wounds, extensive sores or ulcerative lesions on the face. The patient is hypersensitive to any component of the drug.
WARNINGS AND PRECAUTIONS:
Gentian violet can cause irritation and ulceration of the mucous membranes when applied. Avoid direct contact with eyes. Pregnant and lactating women.
USE FOR PREGNANCY AND LACTATION WOMEN:
Caution should be exercised when using the drug for pregnant and lactating women.
Effects on the ability to drive and operate machinery:
There is no evidence of an effect on the ability to drive or operate machinery.
Drug interactions and incompatibilities:
a) Drug interactions: So far there has been no reported information on drug interactions.
b) Drug incompatibility: Since there are no studies on drug compatibility, this drug should not be mixed with other drugs.
Undesirable effects of the drug:
The drug may cause irritation, or cause hypersensitivity reactions and mucosal ulcers. When applied the skin has a blue-purple color. Immediately inform your doctor or pharmacist of any adverse reactions experienced when using the drug.
Overdose and treatment:
There is no data on drug overdose, do not exceed the indicated dose of the drug.
Pharmacological information:
MILIAN solution consisting of Methylene Blue and Gentian Violet is an antiseptic/microbial killer for topical application to the skin and mucous membranes. Methylene blue has irreversible bonds with viral nucleic acids and breaks down the virus’s molecule when exposed to light. Gentian violet is used in the treatment of microbial infections of the skin and mucous membranes caused by Candida albicans, such as oral candidiasis, vaginal, interstitial rash and perinail inflammation.
PRESENTATION:
Box of 1 bottle of 20 ml.
Storage:
Cool and dry place, temperature below 300 C, protected from light. Close the lid immediately after use.
SHELF LIFE:
24 months from date of manufacture.
MANUFACTURER RESPONSIBLE FOR THE GOODS:
OPC Pharmaceutical Joint Stock Company (1017 Hong Bang, Ward 12, District 6, Ho Chi Minh City).
MADE IN VIETNAM.